
Strained Rings
@strained_rings
Strain chemistry insights | Recent papers, perspectives & breakthroughs | Where molecular stress meets discovery 🔬
Handled by @HariGroupIISc
ID: 2007795319132819457
04-01-2026 12:46:47
23 Tweet
408 Followers
88 Following


🔥 Direct Synthesis of complex tricyclo[3.2.1.02,7]octenes via Pd catalyzed Sequential Reactions of 2-Pyranones with strained methylene cyclopropanes. J Org Chem/Org Lett pubs.acs.org/doi/10.1021/ac…

💡Ring-constraint meets photochemistry: photoinduced energy transfer unlocks oxazolones for selective cycloadditions with strained vinyl cyclopropanes and alkenes. A smart strain-enabled route to complex heterocycles. 🧩 J. Am. Chem. Soc. Brown Lab Indiana University pubs.acs.org/doi/10.1021/ja…

🧪 Photocatalytically generated 1,3-dielectrophiles enable [2+1] and [2+1+1] cyclizations of alkenes, forging highly strained small carbocycles with precision. A sleek strategy to access cyclopropanes & beyond 🔬💥 J. Am. Chem. Soc. Seoul National University pubs.acs.org/doi/10.1021/ja…


Brønsted acids trigger strain-release exo-cyclization of bicyclo[1.1.0]butyl ketones to access 2,3-diazabicyclo[3.1.1]heptenes 💥🔬— elegant ring expansion via relief of BCB strain for complex heterocycle synthesis 🧪🔥 J Org Chem/Org Lett pubs.acs.org/doi/10.1021/ac…

🌟 H-bond orchestration + strain-release drives a photochemical 3-component union of alkenes, p-quinone methides & NH₄SCN to form strained spiro-cyclobutanes – leveraging spiro strain for elegant bond construction! J Org Chem/Org Lett Chandra Bhushan Tripathi pubs.acs.org/doi/10.1021/ac…

Strain-release multicomponent chemistry of azabicyclo[1.1.0]butanes drives asymmetric access to quaternary N-α-bisaryl azetidines 🎯– harnessing ring strain for modular, enantioselective azetidine assembly! J Org Chem/Org Lett IIT Kharagpur pubs.acs.org/doi/10.1021/ac…

The first collaborative effort between Santanu Mukherjee and The Hari Group resulted in Chemodivergent Catalytic Enantioselective Ring Opening of Azabicyclobutanes: chemrxiv.org/doi/full/10.26… Kudos to Arko Seal, SUPARNAK MIDYA, and Snehasis Pal for their hard work. IISc Bangalore


⚡ Visible light + organic photocatalyst/CBr₄ unlocks [3 + 2] cycloadditions of strained vinylcyclopropanes with alkenes/alkynes: harnessing ring strain for elegant access to cyclopentanes & cyclopentenes J Org Chem/Org Lett pubs.acs.org/doi/10.1021/ac…

📚Check out the review on "Carbonylative Construction and Transformation of Strained Carbocycles" ACS Catalysis pubs.acs.org/doi/10.1021/ac…

💥⚡Rhodium-catalyzed sulfonylation of strained gem-difluorocyclopropanes delivers regio- & enantioselective fluorinated sulfones, harnessing ring-strain release for elegant C–C/C–F activation and stereocontrol 📐 ACS Catalysis pubs.acs.org/doi/10.1021/ac…

🦋 Visible-light & Lewis acid unlocks diradical H-atom transfer in strained bicyclo[1.1.0]butanes, enabling C(sp³)–H functionalization and cyclization to access 3-azabicyclo[3.1.1]heptanones⚡An outstanding work by Yang XIONG & Israel Fernández Group J. Am. Chem. Soc. pubs.acs.org/doi/10.1021/ja…

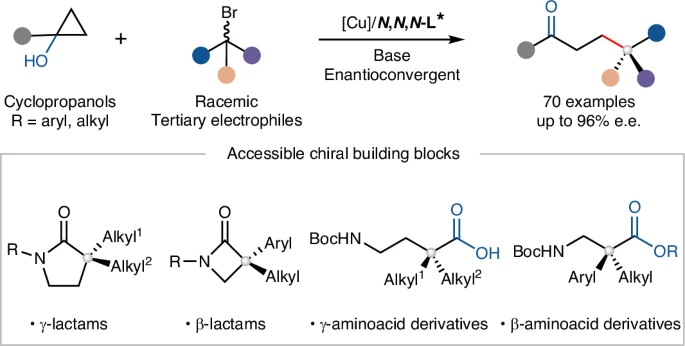
🎯 Cu-catalyzed enantioconvergent radical coupling of strained cyclopropanols with tertiary electrophiles forges quaternary stereocenters, exploiting ring-strain release to access 3D-rich chiral scaffolds 💥 Nature Synthesis rdcu.be/e3WMb


🦋 Strain-release-driven intermolecular (5+3) cycloaddition of bicyclo[1.1.0]butanes with oxidopyrylium delivers architecturally rich bridged frameworks, exploiting extreme BCB strain for rapid polycyclic scaffold construction 💥🔬✨ J Org Chem/Org Lett pubs.acs.org/doi/10.1021/ac…

🦋 Yb(OTf)₃ catalyzes strain-release [2π+2σ] cycloaddition of bicyclo[1.1.0]butanes with methylenimines, delivering richly substituted 2-aza-bicyclo[2.1.1]hexanes—an elegant gateway to 3D N-heterocyclic bioisosteres 💥🔬✨ J Org Chem/Org Lett pubs.acs.org/doi/10.1021/ac…

🔺 Harnessing cyclopropene strain via visible-light radical [4+1] annulation to forge benzofulvenes—an elegant merger of photoredox and strain-release chemistry 🌟 J Org Chem/Org Lett pubs.acs.org/doi/10.1021/ac…

🦋 Photocatalytic reconstruction of ultra-strained bicyclo[1.1.0]butanes unlocks oxygenated bicycles via strain-release reactivity—expanding 3D chemical space with elegance and precision. ⚡🔗🧪 J. Am. Chem. Soc. Hong Group (Medisyn Lab) pubs.acs.org/doi/10.1021/ja…
