Bruno A. Costa, MD
@mdbrunocosta
Internal Medicine Resident @MountSinaiNYC, Research Affiliate @MSKCancerCenter, Myeloma & Supportive Oncology Enthusiast, First-Gen Doc 🇧🇷
ID:1487770644477530121
https://www.ncbi.nlm.nih.gov/myncbi/bruno.almeida%20costa.1/bibliography/public/ 30-01-2022 12:52:33
1,4K Tweets
901 Followers
852 Following


Yesterday, in a ‘Historical’ Moment, FDA Committee Ruled on a Faster Way to Approve Therapies for Myeloma.
We were honored to present EVIDENCE meta-analysis, work I pioneered 15 yrs ago. Congrats also to I2TEAMM!
International Myeloma Foundation Multiple Myeloma RF #mmsm HealthTree Foundation Sylvester Comprehensive Cancer Center


Real world data on bridging therapy for CAR-T. Report by U.S. Myeloma Immunotherapy Consortium Aimaz Afrough Yi Lin Surbhi Sidana, MD Gurbakhash Kaur Larry Anderson,MD,PhD,FACP Shonali Midha, M.D. Hamza Hashmi Krina Patel Douglas W. Sborov MD MS Omar Nadeem et al.
nature.com/articles/s4140…


IMF celebrates ODAC's historic decision on MRD testing as an early endpoint for accelerated myeloma drug approval. 10 yrs of IMF research led by Dr. Durie & i2TEAMM; & U of Miami Sylvester Comprehensive Cancer Center contributed to this decision. Bruno Paiva Nikhil C. Munshi, MD


ODAC votes 12-0 for MRD as a surrogate endpoint for accelerated approval in myeloma.
Kudos to the i2TEAMM led by Nikhil C. Munshi, MD Brian Durie, M.D. Jesus San Miguel for a monumental effort.
International Myeloma Foundation Yelak Biru
targetedonc.com/view/fda-odac-… Targeted Oncology

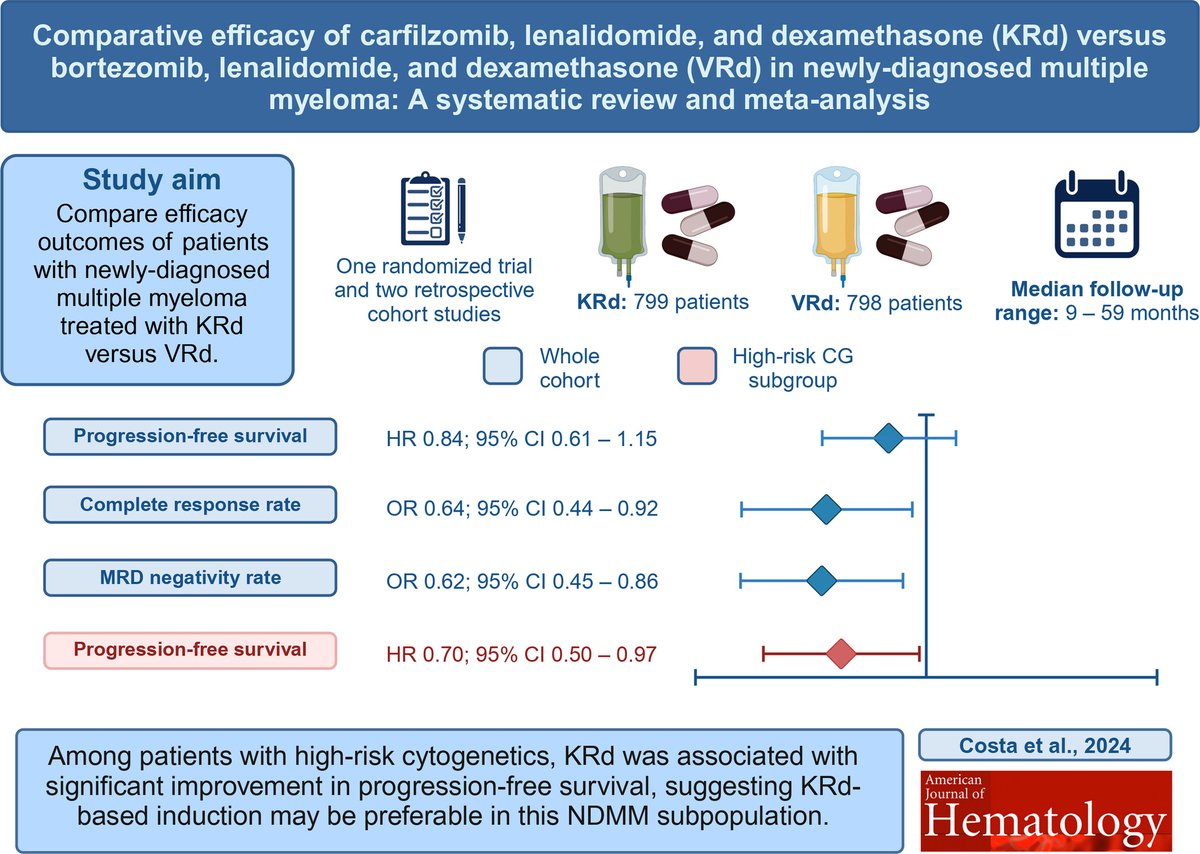
In NDMM, how does KRd compare to VRd regarding efficacy endpoints? Happy to share our new meta-analysis just published in AmericanJournalofHematology: doi.org/10.1002/ajh.27….
KRd-treated pts showed higher odds of achieving MRD negativity and CR/sCR compared to VRd-treated pts, while ORR,…

Using ePROs to prompt direct patient trial invitations resulted in greater enrollment to a symptom-intervention trial than standard of care physician-based referral. ja.ma/4cU8lin Nicholas Verdini Jun J. Mao Erin Gillespie, MD MPH Memorial Sloan Kettering Radiation Oncology





IMO: MRD is a premature test, and using it as an endpoint surrogate in myeloma might be a setback to the field, especially since we don’t have strong evidence that it correlates with OS improvement yet!
#mmsm #myeloma #MedEd #USMIRC USMIRC News FDA Oncology #medtwitter

From Joshua Richter, MD, FACP in Blood Cancer Journal: Like a bridge over troubled water: keeping the myeloma down en route to CAR-T
rdcu.be/dEAj6
#MultipleMyeloma


⬇️ A primer on #ArtificialIntelligence for everyone involved in #CancerResearch to add to their “must read” list!
aacrjournals.org/cancerdiscover…
Cancer Discovery AACR Elizabeth McKenna Eli Van Allen #AACR24


Linvoseltamab Elicits Deep Responses in Late-Stage R/R Multiple Myeloma onclive.com/view/linvoselt… via @onclive with Sundar Jagannath Icahn School of Medicine at Mount Sinai #MultipleMyeloma



New #guideline on Vaccinations for Adults with Cancer are out in Journal of Clinical Oncology! A brief 🧵... 1/
Key Takeaway: Optimize vax status as a vital part of cancer care! Assess status early, vaccinate before treatment when possible, coordinate w/ PCPs & team members. ascopubs.org/doi/10.1200/JC…


CD70-Targeted Allogeneic CAR T-Cell Therapy for Advanced Clear Cell Renal Cell Carcinoma aacrjournals.org/cancerdiscover… MD Anderson Cancer Center