Alexey Aleshin
@aaleshin
Oncologist. Researcher. Corporate CMO and GM of ECD at Natera. Helping bring ctDNA testing to patients. Opinions my own
ID: 36709019
30-04-2009 16:48:02
116 Tweet
293 Takipçi
147 Takip Edilen

Disappointed. Despite AZ, IL, LA, RI—recently passing legislation to expand access to #BiomarkerTesting, Governor Gavin Newsom vetoed this bill #SB912, despite unanimous bipartisan support. Big step backwards for CA patients and doctors. #CallOnNewsom #ctdna genomeweb.com/policy-legisla…




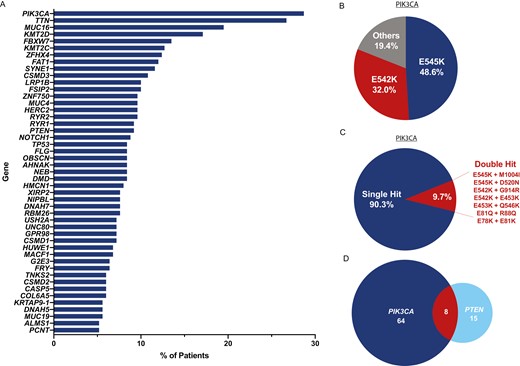
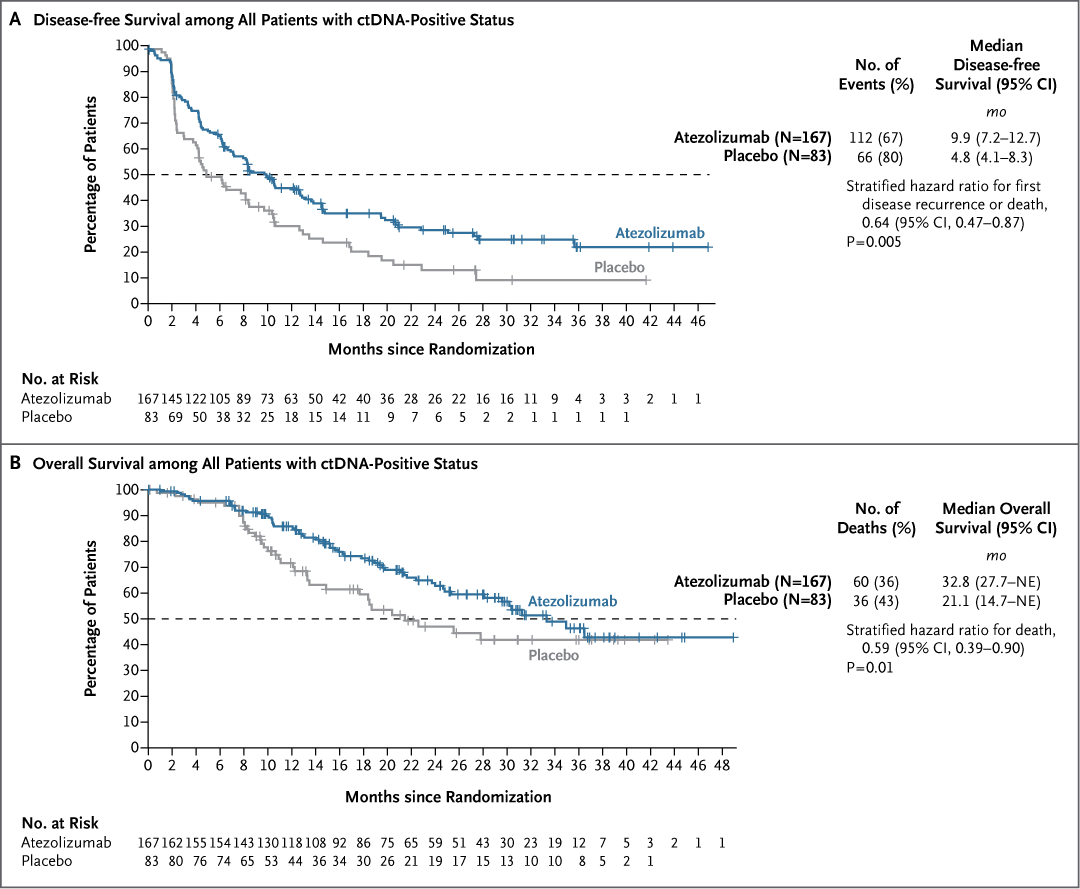
Updated Overall Survival by Circulating Tumor DNA Status from the Phase 3 IMvigor010 Trial buff.ly/3O6YHxr Tom Powles Zoe Assaf Petros Grivas Alexey Aleshin joaquim bellmunt Sanjeev Mariathasan



Patients with colon cancer with positive ctDNA tests often face tougher outcomes. Research by Dana-Farber & Brigham and Women's Hospital presented at #ASCOGI shows that treatment with a non-steroidal anti-inflammatory drug significantly boosts disease-free survival. ➡️ bit.ly/4g9zO01


ctDNA Status Is Prognostic for DFS With Celecoxib in Stage III Resected Colon Cancer Jonathan A. Nowak Brigham and Women's Hospital Brigham and Women's Pathology Dana-Farber Harvard Medical School ASCO #GI25 #crcsm #medtwitter #oncology onclive.com/view/ctdna-sta…

CALGB SWOG 80702 results to be presented today. First large scale predictive Signatera data in CRC showing benefit to adding celecoxib to SOC ACT in S3 disease. Big news for field, congrats to all involved. Luis Natera, Ph.D. #alliance #GI24 #ctdna $ntra natera.com/company/news/n…

VIDEO: Celecoxib / Celebrex & ctDNA in Stage III Colon Cancer findings from the CALGB Alliance / SWOG 80702 at ASCO GI Jonathan A. Nowak ASCO #GI25 Brigham and Women's Hospital Brigham and Women's Pathology Dana-Farber Harvard Medical School OncoAlert #Celecoxib #ctDNA #ColonCancer #ASCOGI2025 #CancerResearch #Oncology