Scott Vafai, MD
@scottvafai
SVP of Clinical Development @VerveTx. Formerly @sanofi, @broadinstitute, @MassGeneralNews, @BrighamWomens. All opinions are my own.
ID: 716339346501197824
02-04-2016 18:59:43
274 Tweet
468 Takipçi
1,1K Takip Edilen

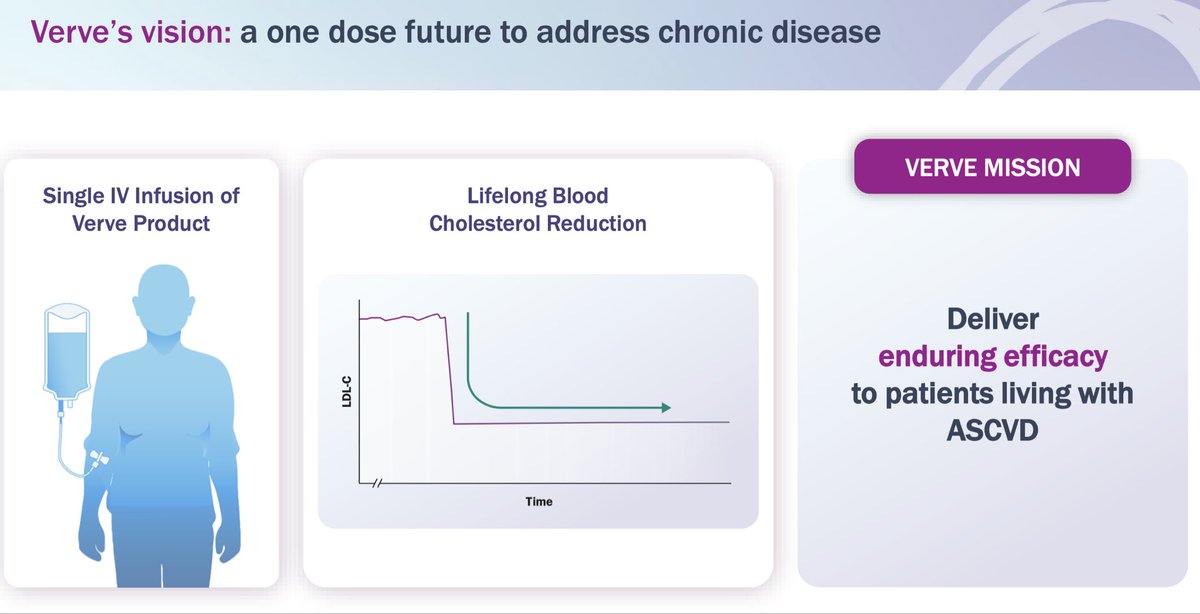
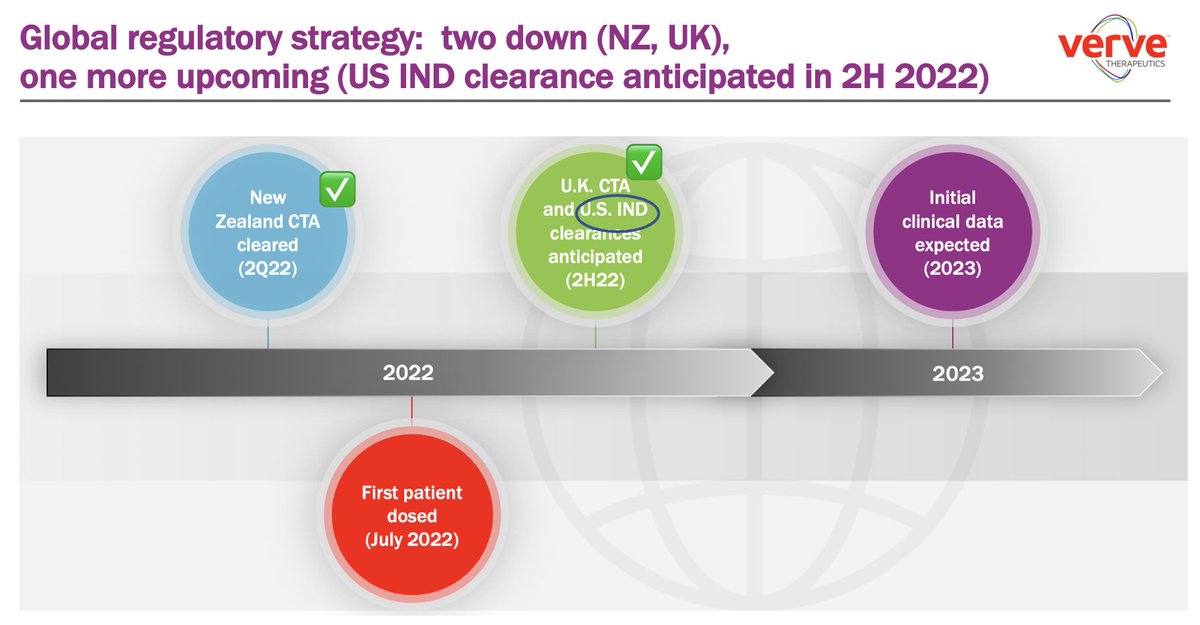
2018: Verve Therapeutics founded with concept of one-time Rx to lower LDL as low as possible for as long as possible Today: we are announcing dosing of *first patient* with VERVE-101, an in vivo CRISPR base editing medicine 🙏 to patient volunteer $VERV globenewswire.com/news-release/2…


👇🏽 Heard on the Street from David Wainer Appreciate the opportunity to share the @VerveTx story The Wall Street Journal $VERV


Excited to announce today: development candidate for 2nd program (VERVE-201 targeting the ANGPTL3 gene) to treat homozygous FH as well as ASCVD data to be presented by Amit V. Khera European Society of Cardiology 2022 Press release: ir.vervetx.com/news-releases/… Data: ir.vervetx.com/static-files/3… $VERV


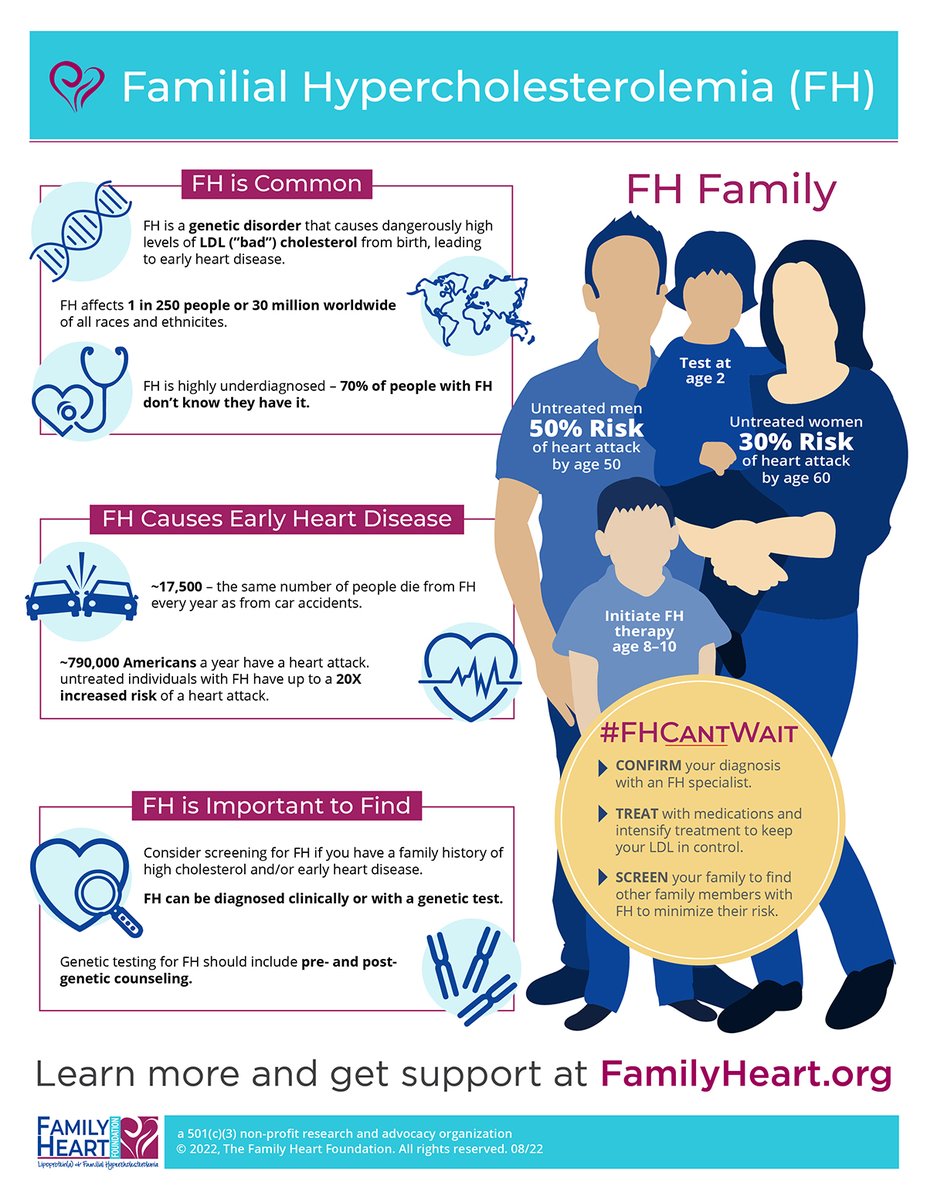
Delighted to announce a Clinical Trial Authorization (CTA) clearance by UK MHRA (Medicines and Healthcare products Regulatory Agency) for VERVE-101 as investigational Rx for patients with heterozygous familial hypercholesterolemia ir.vervetx.com/news-releases/… $VERV Verve Therapeutics



For @VerveTx targets, 2 criteria: human genetics & human pharmacology VERVE-201 targets ANGPTL3 Human genetics (L): complete deficiency ANGPTL3 = low blood lipids, no liver fat New human pharmacology (R): siRNA ArrowheadPharma, mimics human genetics! researchsquare.com/article/rs-209…


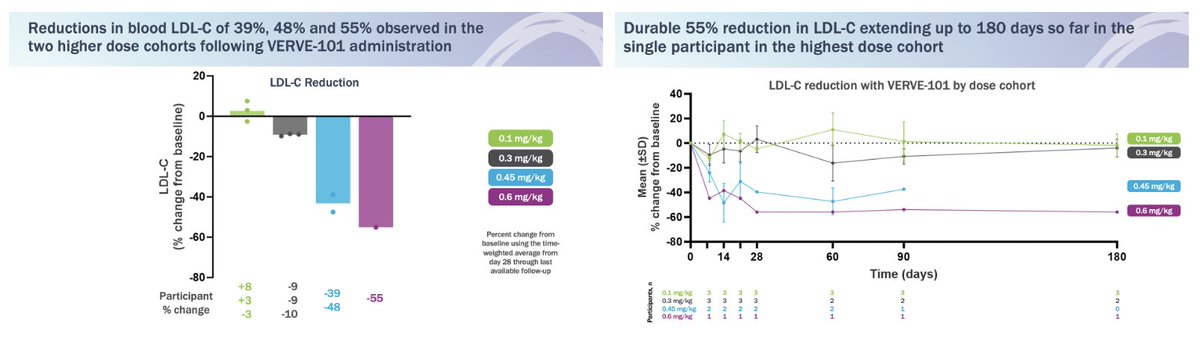
In Circulation, we publish today pre-clinical data for VERVE-101 Of note, American Heart Association tells us that for Scientific Sessions '22, this abstract the highest ranked one of all submitted from U.S. #AHA22 Press release: globenewswire.com/news-release/2… Paper: ahajournals.org/doi/10.1161/CI…


👇news Verve Therapeutics and Eli Lilly and Company establish global collaboration to advance an in vivo gene editing program targeting Lipoprotein(a) for the treatment of atherosclerotic cardiovascular disease ir.vervetx.com/news-releases/…



We are thrilled to gain Eli Lilly and Company as a new partner for @VerveTx PCSK9 & ANGPTL3 programs👇 Lilly has purchased from Beam Therapeutics all of their previously held product rights in PCSK9 & ANGPTL3 programs for a $600M total potential deal value. For the PCSK9 & ANGPTL3 product





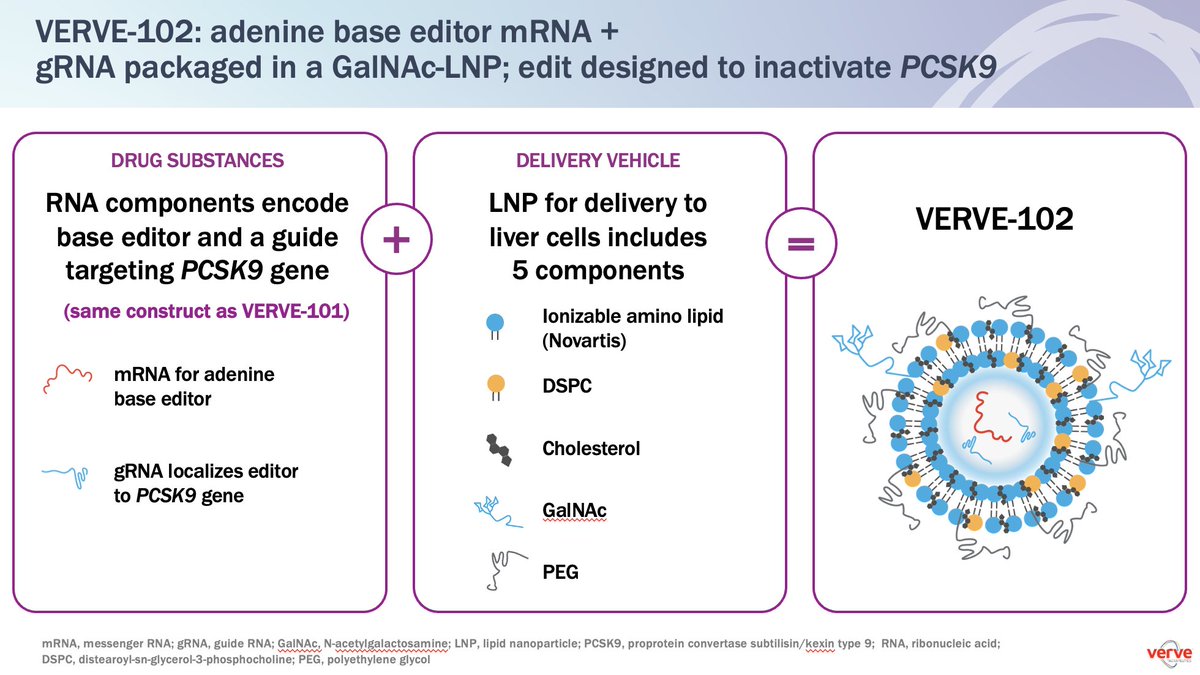
Glad to announce the dosing of the first patient with VERVE-102 in the Heart-2 clinical trial VERVE-102: *in vivo base editor to permanently turn off the PCSK9 gene in liver and durably lower blood LDL-C *delivered using Verve Therapeutics proprietary GalNAc-LNP delivery system as a



We are hiring! Delighted to share an opportunity to join the clinical development team at Verve Therapeutics Eli Lilly and Company , where we are on a mission to transform the care of ASCVD through single-course gene editing medicines! Apply here: lnkd.in/eVDDP85i