Nicole McCabe
@NicoleALSJurney
35 year old PNW girl battling #ALS I'm here to fight with all my might. My babies need me! We deserve a right to live. 💙 join me in my battle
ID:541446126
31-03-2012 04:36:53
6,8K Tweets
1,5K Followers
650 Following



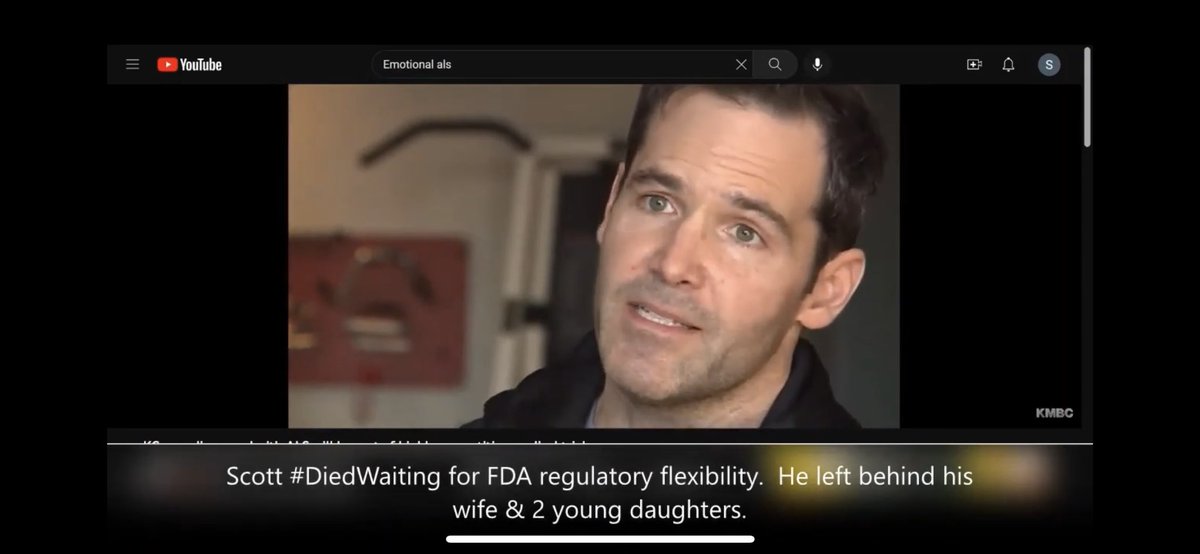
Dr. Robert M. Califf What is more terrible is U.S. FDA FDA Biologics blocking and delaying safe and effective treatments for 100% fatal disease #ALS ( #NurOwn ) with its stodgy process. There have been thousands begging for access 8+ years but are stymied and left #DiedWaiting on FDA
wlwt.com/article/local-…

Day 28 of ALS Awareness month - PART 1! If you want to help, call your congressperson and recommend Nurown’s FDA approval. #als #als awarenessmonth #lougehrigsdisease #neurology #fda #clinicaltrial #endpoint #nurown #treatment #medicalnews #congress #advisorycommittee #adcom


. Dr. Robert M. Califf FDA Biologics
2015 #NurOwn works in P2 - K no sx
2019 #NurOwn Works in P3 - K dx'd #ALS
2023 Still no ADCOM while people who rcvd doses in EAP outliving natural history
Young mom's suffering =
Real life impact of Type II error & lack of urgency
#ALS AwarenessMonth
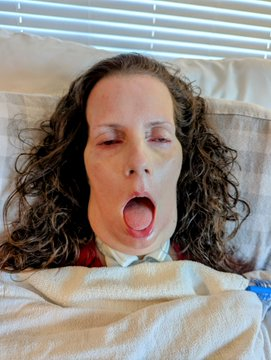

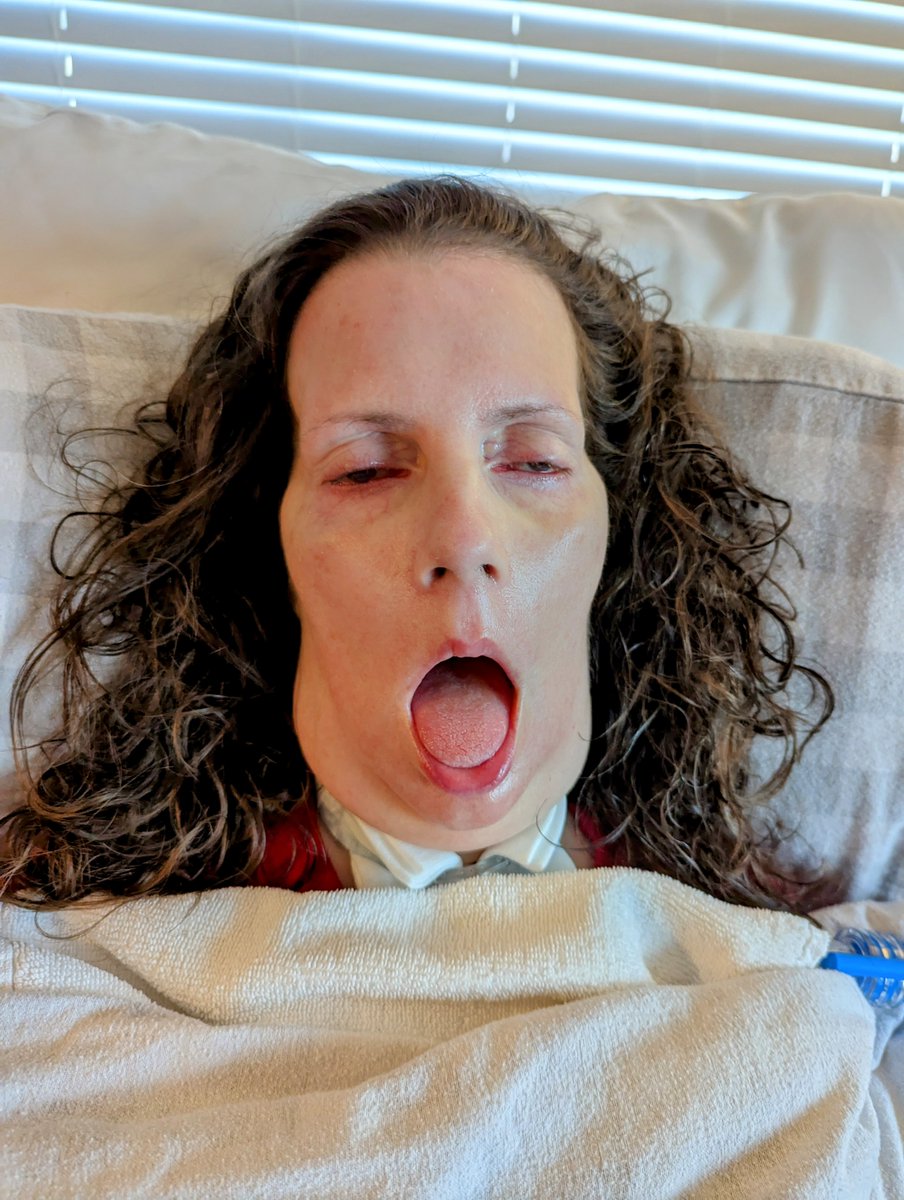
Kristin is dying of #ALS .
You're MISTAKEN if you believe that HORRIFIC 100% FATAL ALS can't happen to YOU.
CONGRESS, DEMAND that FDA Biologics uses regulatory flexibility for this disease WITH NO SURVIVORS.
Dr. Robert M. Califf, grant NUROWN Accelerated Approval with a Phase 4

One would never know that #ALS is a rapidly 100% FATAL disease by this timeline.
12/27/22-FDA Biologics grants Nurown Type A mtg
1/11/23-date of Type A mtg
3/27/23- #FDACBER grants AdComm hearing
5/2/23-ALS patients #DyingWaiting on FDACBER announcement of AdComm hearing date...
If

23-year-old Jacob below has #ALS .
WE NEED CONGRESS. After broken promises of regulatory flexibility, several generations of ALS #DiedWaiting despite YEARS of #NUROWN safety & UNPRECEDENTED #RealWorldEvidence
Give dying ALS HOPE. DEMAND that FDA Biologics grant NUROWN Accelerated


Stevens family credits Eric's condition to #NurOwn
'Not many patients can say they're stil walking, talking & eating almost 4 yrs into diagnosis... Eric is #LivingProof that experimental therapies can work.'
Asking FDA Biologics to listen to Eric's #RWE
dailybreeze.com/2023/04/05/for…


Jacob below has #ALS diagnosed at 23 years old.
Dying ALS have broken bodies & no Big Pharma allies
WE NEED CONGRESS to DEMAND the FDA Biologics #ApproveNurown .
END the CRUEL regulatory RIGIDITY for a disease WITH NO SURVIVORS since 1869
Give Jacob & ALL with 100% fatal #ALS HOPE.


Today, we are another step closer to ending ALS.
President Biden's budget request includes $2.5 million to implement #ACTforALS to foster the development of treatments for ALS and other rare neurodegenerative diseases.

Today, for the first time since last winter, Chris Snow put on his skates. He had to get used to a whole new body since the last time he was on the ice — one with 2 almost useless arms — but he did it & told me during this kids v parents game, “This is so fun.” #nevergiveup

Shortly after his diagnosis, Brian Wallach and Sandra came to my office to discuss reforming a healthcare system not designed for a disease like ALS.
A little over a year ago, we saw Act for ALS signed into law.
Together, we will find treatments and a cure to end ALS.


Wes, diagnosed with 100% FATAL #ALS at 33 years old, died leaving behind his wife & 2 boys
ALS devastation continues EVERY DAY IN EVERY COMMUNITY
CONGRESS, END the CRUEL regulatory RIGIDITY of the FDA Biologics.
DEMAND Dr. Robert M. Califf grant an AdComm hearing for safe/effective #Nurown



Day 570 asking:
U.S. FDA to use flexible regulatory pathways with BrainStorm’s #NurOwn for #ALS .
&
Dr. Patrizia Cavazzoni President Biden Vice President Kamala Harris Dr. Robert M. Califf to act urgently.
#everypointmatters so that my mom, a #PLWALS , can meet my kids, not just my dog. There remains an unmet need here.


As co-chair of the Rare Disease Congressional Caucus, I’m proud to reintroduce the BENEFIT Act with Rep. Brad Wenstrup that would provide patients and advocates the ability to play a larger role in the U.S. FDA's benefit-risk framework for drug approval.
matsui.house.gov/media/press-re…