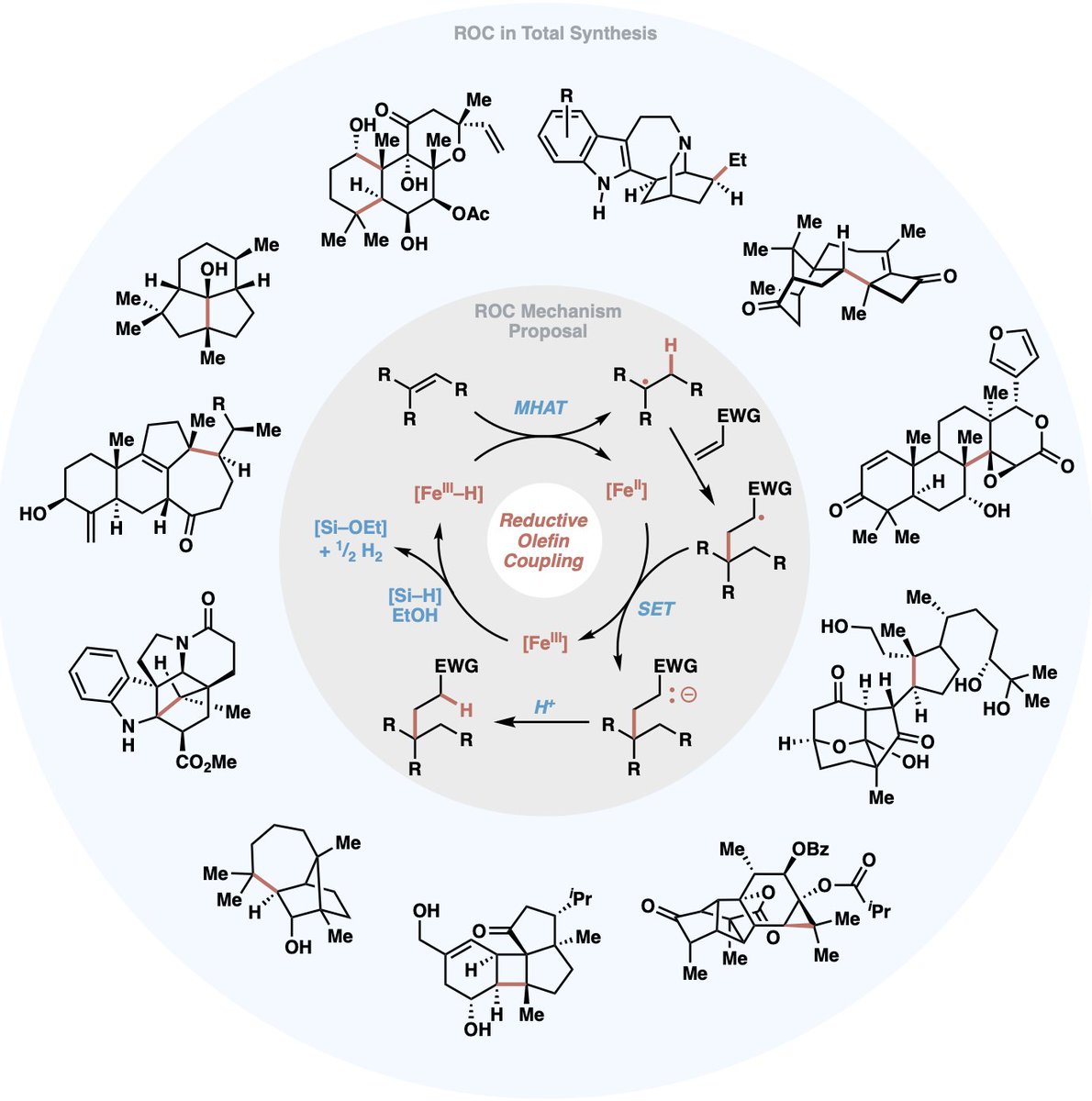
SUBHADEEP HAZRA
@subhadeephazr12
# Researcher
# center of Biomedical Research.
# NIPER Mohali SAS NAGAR
ID: 1419601211951513602
https://jaideepsaha-niper.weebly.com/ 26-07-2021 10:11:27
89 Tweet
44 Followers
172 Following

Arka Porey Nature Chemistry Fantastic work 👏






Our lab UAlberta Chemistry has 2 PhD positions open for Fall 2026 in catalysis and synthetic organic chemistry. Contact me via email if interested!

Pleased to share our recent work published in J. Am. Chem. Soc. pubs.acs.org/doi/10.1021/ja… on N-N cross coupling via nitrene transfer reaction. Discovery of a new nitrene transfer reagent! Thank you Bas Bas de Bruin for this great collaboration. Anusandhan National Research Foundation IISER Pune Chattopadhyay Group

Happy to share our work on a Lewis acid–controlled divergent annulation of o-quinone methides with bicyclo[1.1.0]butanes, enabling selective pathways to sp³-rich oxa-bicyclic frameworks. doi.org/10.1021/acs.or… ACS Publications J Org Chem/Org Lett Soumik Mondal MANVEER PATEL

We’re excited to introduce a Twitter account Strained Rings dedicated to strained‑ring chemistry—your go-to source for cutting-edge papers, fresh perspectives, and breakthrough discoveries in ring strain chemistry. Please follow for updates! x.com/strained_rings…



Try to use simple light and Phth-SCF3 to activate your amine... Scuola di Scienze Unipd Nature Communications Università di Padova Dipartimento di Scienze Chimiche - UniPD


Want to learn more about our strained ring chemistry? Jerome will give a talk at the Thieme Cheminar Strain-Release Chemistry on February 24, 11-13 CET. Hosted by Prof. Biju and featuring also presentation of The Hari Group and Wipf Group . cassyni.com/events/P3TyBNe…

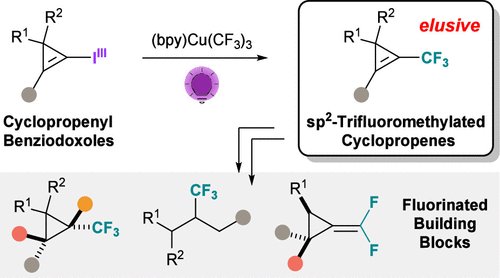
Very excited to join Waser Group & The Hari Group at the Cheminar on Strain-Release Chemistry ⚡️⚡️⚡️ cassyni.com/events/P3TyBNe…


Happy to share our recent work HFIP-promoted strain-release functionalizationof Azabicyclo[1.1.0]butane enables rapid access to medicinally relevant 3,3-disubstituted oxetane, azetidine & thietane scaffolds via trichloroacetimidate activation. doi.org/10.1021/acs.or… J Org Chem/Org Lett

Happy to share a new way of promoting dual-atom insertion into oxetanes and azetidines to access multi-heteroatom rings using FLP chemistry, just out in Nature Synthesis. nature.com/articles/s4416… #skeletalediting #ringexpansion #FLP


Excited to share our work! Dual strain-release chemistry enabling rapid azetidine synthesis. Where strain meets strain ⚡ J Org Chem/Org Lett doi.org/10.1021/acs.or…

🦋 Double strain, single strategy 💥! Lewis acids unite azabicyclo[1.1.0]butanes with D–A cyclopropanes/BCBs, converting extreme ring strain into richly functionalized azetidines—compact rings → complex 3D space ⚡ J Org Chem/Org Lett Jaideep Saha pubs.acs.org/doi/full/10.10…

Our latest work in Organic Letters "Electroreductive Cross-Coupling of Cyclopropyl Ketones and Aryl Methyl Ketones to Access 1,2,4-Triarylbenzenes via Acid-Promoted Aromatization " We are thankful to IIT ROPAR J Org Chem/Org Lett DSTIndia and congrats to Rohan Bag pubs.acs.org/doi/10.1021/ac…

![Dell'Amico Group (@dellamico_group) on Twitter photo Want to put a bit of sweetness in your week ?? 🍰
Check out our new paper on a radical strategy to synthesize bicyclo[1.1.1]pentyl C-glycosides !!
Thank you to all the authors and collaborators for this work !
urlr.me/Pnwbj2 Want to put a bit of sweetness in your week ?? 🍰
Check out our new paper on a radical strategy to synthesize bicyclo[1.1.1]pentyl C-glycosides !!
Thank you to all the authors and collaborators for this work !
urlr.me/Pnwbj2](https://pbs.twimg.com/media/G59DpIxXAAAxRDb.png)