NEW FDA DRUG APPROVAL
Medicine: Saphnelo
Active Ingredient: Anifrolumab
Indication: Moderate-to severe systemic lupus erythematousus along with standard therapy
Dosage Form: 300 mg/2 mL in a single-dose vial
Mechanism: Type I interferon receptor antagonist
#TopUpPharmacy



Thank you Dr. Winkler & AstraZeneca for a great presentation on Saphnelo. DYK? #Ultrasound evaluates msk manifestations of SLE? Detect Tenosynovitis > synovitis, monitor treatment response #rheumtwitter #lupus #arthritis #POCUS #RhMSUS






Grace Wright MD PhD explains the importance of the interferon pathway in pts suffering from #lupus #SLE
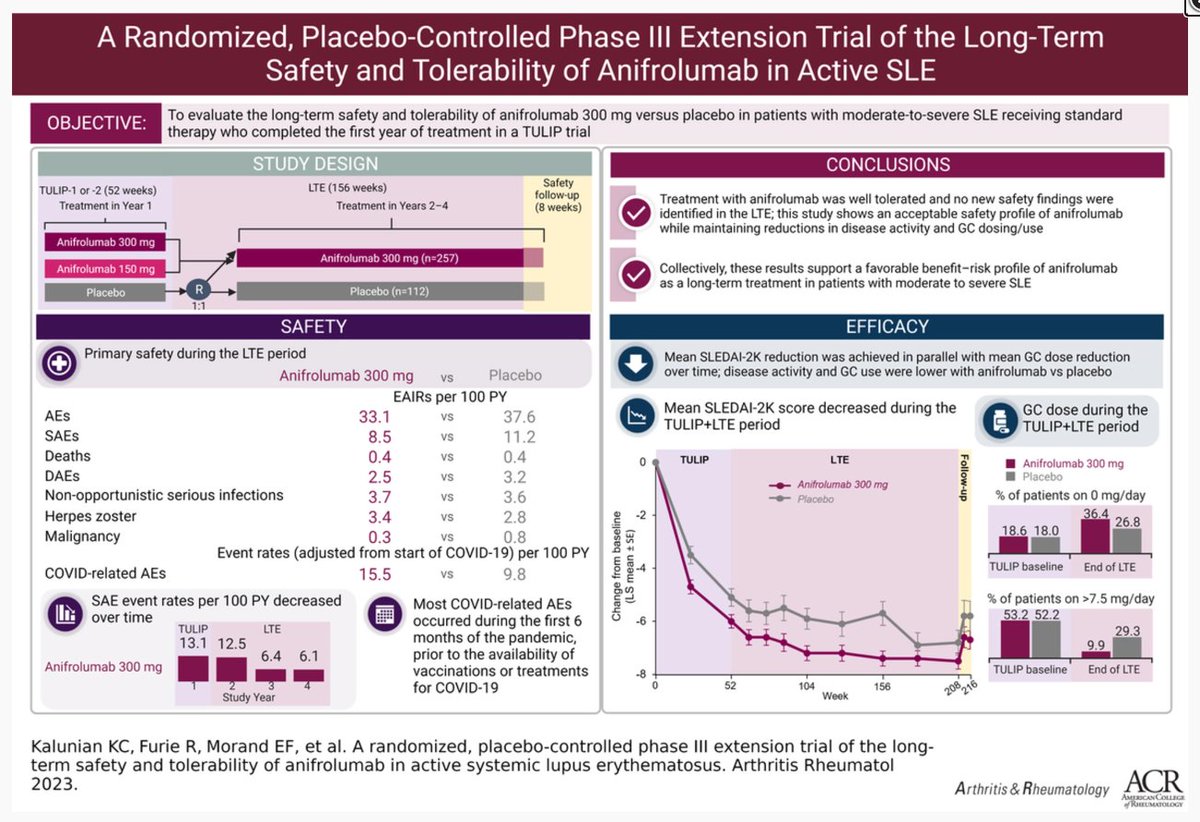
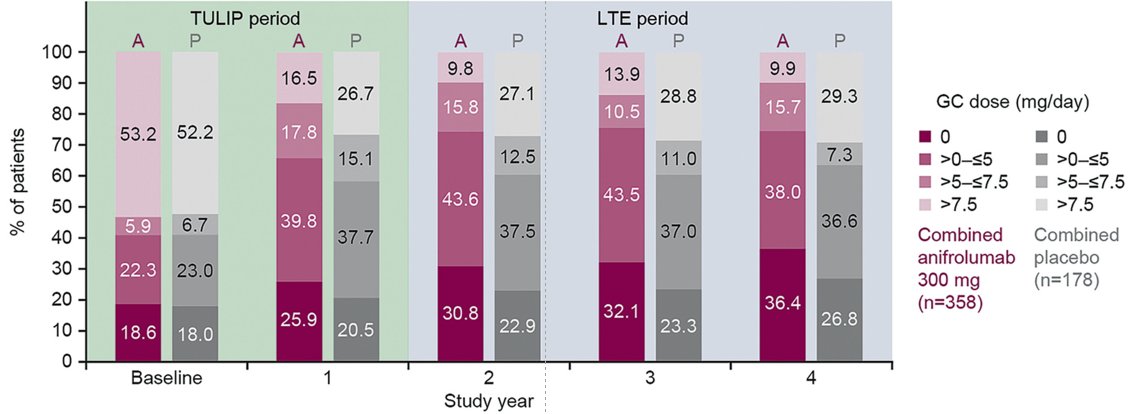
In TULIP2, @Saphnelo is
-effective in multiple organs (BICLA, CLASI)
-steroid-sparing (52% v 30% achieved pred<7.5)
-diverse across pt populations
AstraZenecaUS Association of Women in Rheumatology



Thank you to our sponsor AstraZenecaUS @AstraZeneca for your support of #ultrasound education
#MSUS #meded #lupus SAPHNELO® (anifrolumab-fnia)




Based on my lab work it doesn’t look like the #saphnelo is working…… my inflammation rates are just going up #chroniclife #chronicpain