
Pauline Richaud
@pauline_richaud
#PR rep, #NYC addicted, red #wine lover, talk to me about #USA, #health #WorkingMum and #biotech and I'll be #happy! ex @TBWACORPORATE ex @sopexa_agency
ID: 91075821
https://www.linkedin.com/in/paulinerichaud/ 19-11-2009 10:18:53
2,2K Tweet
382 Takipçi
2,2K Takip Edilen



📅 [#EVENT] – Join Alexandre Kiss, Europe Medical Advisor for Oncology and Hematology at MaaT Pharma, at the 17th “Journée Greffe” of Institut Paoli-Calmettes (IPC), taking place on January 25, in Marseille, France. This annual event has become a key moment to reflect on and shape the
![MaaT Pharma (@maat_pharma) on Twitter photo 📅 [#EVENT] – Join Alexandre Kiss, Europe Medical Advisor for Oncology and Hematology at MaaT Pharma, at the 17th “Journée Greffe” of <a href="/paoli_calmettes/">Institut Paoli-Calmettes (IPC)</a>, taking place on January 25, in Marseille, France.
This annual event has become a key moment to reflect on and shape the 📅 [#EVENT] – Join Alexandre Kiss, Europe Medical Advisor for Oncology and Hematology at MaaT Pharma, at the 17th “Journée Greffe” of <a href="/paoli_calmettes/">Institut Paoli-Calmettes (IPC)</a>, taking place on January 25, in Marseille, France.
This annual event has become a key moment to reflect on and shape the](https://pbs.twimg.com/media/GiDc0ErXMAAvvJm.jpg)

The phase 3 ARES study, evaluating MaaT013 in patients with acute GVHD with gastrointestinal involvement after treatment refractoriness to steroids or ruxolitinib, met its primary end point of GI-ORR at day 28. Mohamad Mohty #hematology #oncology onclive.com/view/ares-tria…


💡 What does recent data tell us about the potential for targeting #aGvHD through the #microbiome? ICYMI, learn more about our Phase 3 results from Professor Mohamad Mohty:

📰 – Proud to keep sharing the positive Phase 3 data in #aGvHD through media coverage, this time featured in Scrip, Citeline Commercial! Thanks to Alaric DeArment for reporting on our milestones, including the Phase 3 data and our EMA filing. 👉 Read the full article: insights.citeline.com/scrip/therapeu…



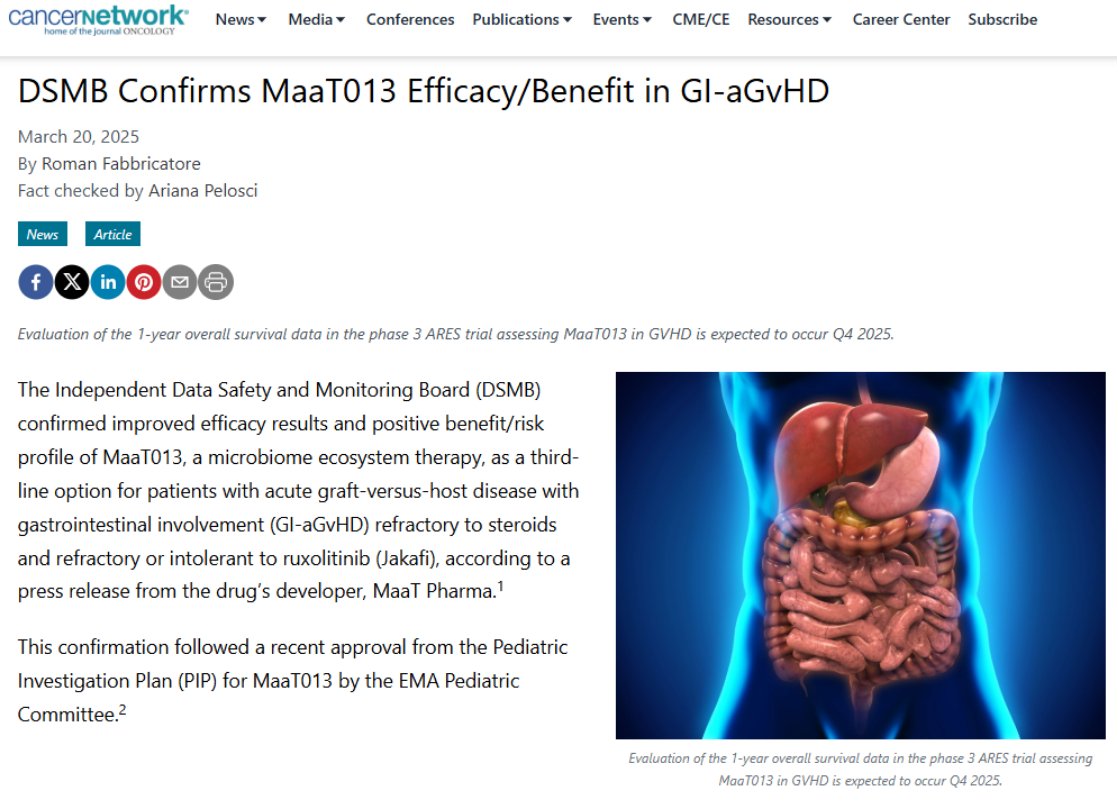
📰 MaaT Pharma in CancerNetwork® ! We’re thrilled to see our latest achievement covered in CancerNetwork® with Roman Fabbricatore highlighting our phase 3 ARES results, confirming MaaT013’s positive benefit/risk profile and its potential as a third-line treatment for GI-aGvHD.


🎥 #EBMT2025 – An insightful interview with Prof. Florent Malard and Prof. Riccardo Masetti on the impact of the microbiome in HSCT outcomes! Prof. Malard discusses how a diverse microbiome correlates with improved survival rates and reduced risk of GvHD, while also mentioning


📰 MaaT Pharma in BioTuesdays! We’re proud to see our CEO, Hervé Affagard Herve Affagard, highlight our pioneering work in Microbiome Ecosystem Therapies, to transform cancer care and improve patient outcomes. A big thank you to Melane Sampson for this great feature in BioTuesdays!



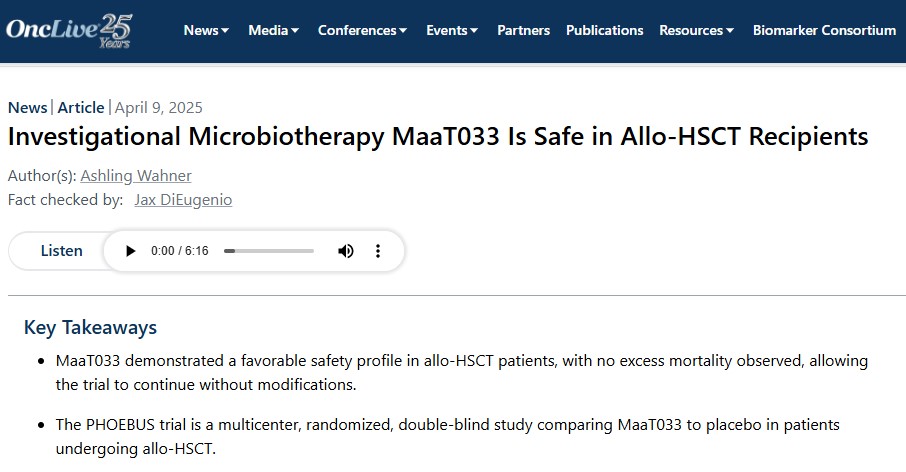
📰 MaaT Pharma in OncLive.com! Thrilled to see our latest announcement covered by Ashling Wahner, highlighting MaaT033’s safety in allo-HSCT patients and the DSMB’s recommendation to continue the PHOEBUS trial. You can read the article here: onclive.com/view/investiga… #DSMB #Safety



Excited to be at #AACR25 and proud to present new promising preclinical data on MaaT034 for the first time! Bastien Laperrousaz, our head of preclinical development, is looking forward to connecting and sharing more about this innovative program!





![MaaT Pharma (@maat_pharma) on Twitter photo 📄 [Communiqué de presse] – MaaT Pharma annonce que le premier patient aux États-Unis a été traité avec MaaT013 à l’hôpital City of Hope dans le cadre du programme d’accès élargi pour la maladie aiguë du greffon contre l’hôte (aGvH).
Ce traitement à usage compassionnel a été 📄 [Communiqué de presse] – MaaT Pharma annonce que le premier patient aux États-Unis a été traité avec MaaT013 à l’hôpital City of Hope dans le cadre du programme d’accès élargi pour la maladie aiguë du greffon contre l’hôte (aGvH).
Ce traitement à usage compassionnel a été](https://pbs.twimg.com/media/GeDkAe0WsAA7wzd.jpg)
![MaaT Pharma (@maat_pharma) on Twitter photo 📄 [Press release] – MaaT Pharma is proud to announce positive Phase 3 results evaluating MaaT013 in treating acute Graft-versus-Host Disease with gastrointestinal involvement (#aGvHD).
Topline Data highlights:
✅ Primary endpoint met: significant GI-ORR at Day 28 of 62%, 📄 [Press release] – MaaT Pharma is proud to announce positive Phase 3 results evaluating MaaT013 in treating acute Graft-versus-Host Disease with gastrointestinal involvement (#aGvHD).
Topline Data highlights:
✅ Primary endpoint met: significant GI-ORR at Day 28 of 62%,](https://pbs.twimg.com/media/GgzDyQuXMAAnQfn.jpg)




![MaaT Pharma (@maat_pharma) on Twitter photo 📰 [#CommuniquéDePresse] - MaaT Pharma annonce quatre présentations lors du 52ᵉ Congrès Annuel de l’EBMT et met en lumière le symposium organisé par Clinigen sur la prise en charge de la GvHD aiguë.
🔹 Présentation orale par le Professeur Florent Malard– Session plénière 📰 [#CommuniquéDePresse] - MaaT Pharma annonce quatre présentations lors du 52ᵉ Congrès Annuel de l’EBMT et met en lumière le symposium organisé par Clinigen sur la prise en charge de la GvHD aiguë.
🔹 Présentation orale par le Professeur Florent Malard– Session plénière](https://pbs.twimg.com/media/HC_KrlaX0AA2nlf.jpg)