mschito
@mschito1
Open to employment.
ID: 1108074544122609664
19-03-2019 18:35:33
125 Tweet
66 Followers
60 Following


Global Remote Research Scholars Program - GRRSP is excited to announce another high value publication ✅ 👇 Authored by Dr.Gowthami_Jagirdhar @doxycyclinitin Rakhtan Qasba @haoirse_ in collaboration with CURE ID Rahul Kashyap, MD, MBA 🇺🇸🇮🇳 mschito 🙏🥳😇


Great to hear David Fajgenbaum, MD's journey from #RareDisease patient to innovator in #DrugRepurposing leading Every Cure. This week, REMEDi4ALL is at the Critical Path Institute (C-Path)'s Drug Repurposing event in the US. It's exciting to share best practices with global peers!


At the Critical Path Institute (C-Path) /cdrc meeting yesterday, c-path.org/april-18-20-20…, I picked up this tip for treating #nausea: aliem.com/trick-trade-is… More sessions on #DrugRepurposing and rare #Cancer today at 1, linked above.

What a great event put on by Critical Path Institute (C-Path)'s CDRC team. As a communicator, I have a lot to learn about the scientific and medical contributions our organization and focus groups make. And I sure did, about the value and importance of drug repurposing. Truly life-saving work.



Deadly Fungal Infections Confound Doctors: ‘It’s Going to Get Worse’ Once a freak occurrence, fungi resistant to standard drugs now threaten millions of Americans With quotes from FungalDoc: moved to bluesky Patrick Bryan Peter G Pappas Tom Chiller George Thompson Fariba Donovan wsj.com/articles/deadl…

Dr. Marco Schito (mschito), Executive Director of CDRC at Critical Path Institute (C-Path), will be a guest lecturer for the Celdara Medical High-Potential Entrepreneurs' Fellowship (CHEF) Program. His lecture will offer valuable insights into challenges & opportunities of drug repurposing.




So exciting to see our podcast on #CUREID and #CDRC featured on Q&A with #FDA about #drug #repurposing for difficult-to-treat #diseases. Podcast available at: fda.gov/media/170476/d… U.S. FDA FDA Drug Information FDA Global FDA Oncology FDAWomen NCATS NIH Critical Path Institute (C-Path)

#CTOS2023 Ultra-rare #sarcomas session underscored the vital importance and urgent need for collaboration among sarcoma communities, highlighting the crucial role of working together in this field. William Tap Liz Connolly Silvia Stacchiotti Anna Maria Frezza CTOS




Critical Path Institute (C-Path) ‘s CURE Drug Repurposing Collaboratory is taking part in the PUSH project (Pushing Ultra-Rare Sarcomas Beyond Hope – International Trial ). We look forward to this collaboration and working on this important project #CTOS2023 #UltraRareSarcoma



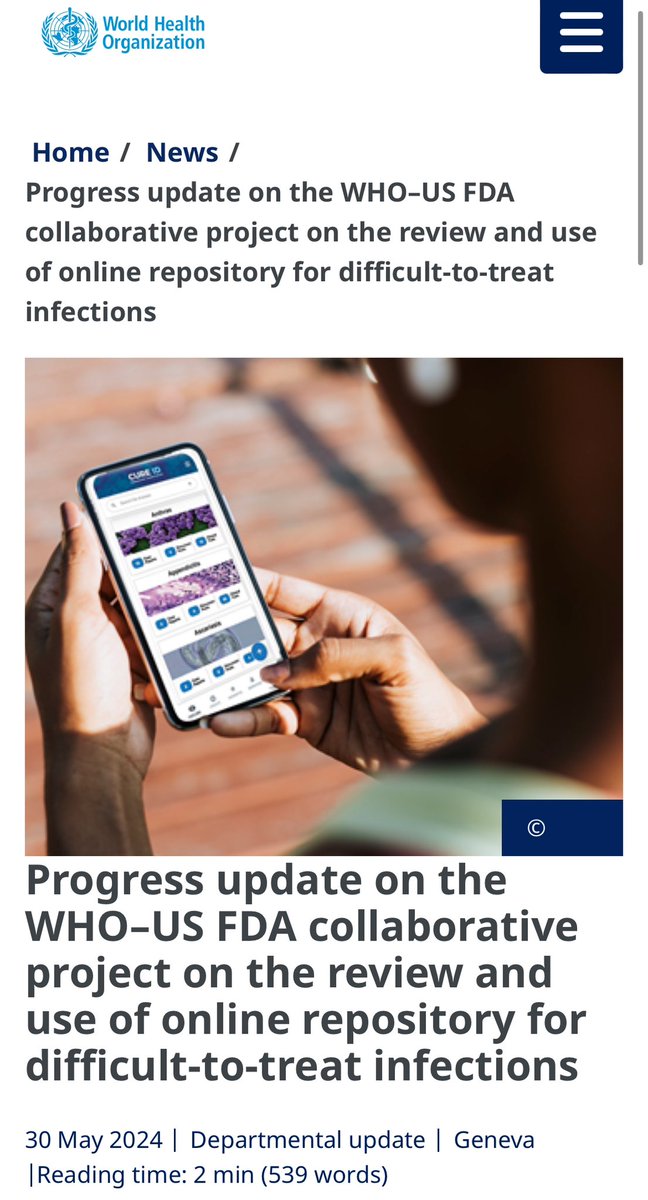
Check out the World Health Organization (WHO)’s recent news about CURE ID titled “Progress update on the WHO–US FDA collaborative project on the review and use of online repository for difficult-to-treat infections” who.int/news/item/30-0…