CellPort Software
@cellportsw
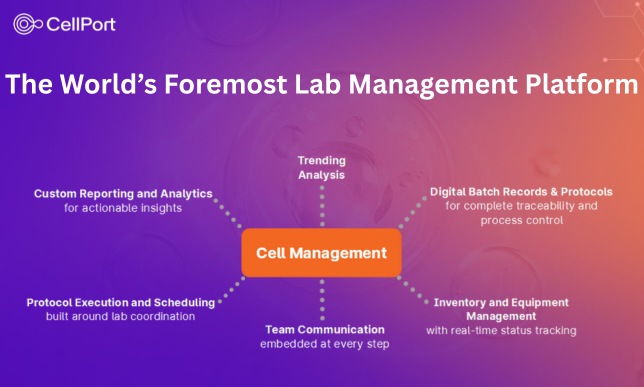
CellPort boasts the most innovative suite of cloud-based cell-manufacturing and lab execution platforms in the life-sciences industry.
ID: 1353519627922501632
http://www.cellportsoftware.com/?utm_source=twitter&utm_medium=profile&utm_campaign=socialmedia 25-01-2021 01:47:01
193 Tweet
26 Takipçi
16 Takip Edilen